Calculator Online
Pages
Category
Follow Us On:
Enter any periodic element and the calculator will instantly calculate the electronic configuration of it along with atomic number, mass number, and state.
Related
An online condensed electron configuration calculator helps you determine the electron configuration of any element. This valence electron calculator displays the abbreviated configuration and atomic number of each element. Read on to understand abbreviated electron configuration, shells, subshells, and how to find the electron configuration of an atom or element.
In quantum chemistry and atomic physics, the electron configuration of an atom or molecule describes the distribution of electrons in atomic or molecular orbitals. It also describes each electron as moving in an orbital within an average field generated by other electrons. For example, the electron configuration of Phosphorus (P) is: 1s² 2s² 2p⁶ 3s² 3p³.
Physicists and chemists often use an electron configuration calculator to determine the electronic configuration of atoms and molecules. The standard notation consists of atomic subshell labels (1s, 2s, 2p, etc.), with the number of electrons in each subshell written as a superscript. Examples:
Electron configuration is also used to:
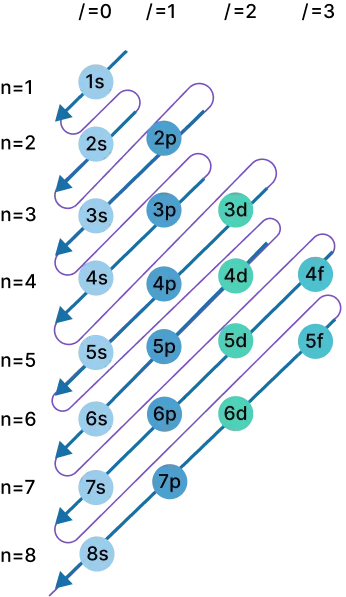
The ground state electron configuration calculator also provides a shorthand (abbreviated) method for finding electron configurations. To do it manually:
Electron shells are sets of allowable states with the same principal quantum number n. The nth shell can hold up to 2n² electrons. For example, the first shell holds 2, the second shell 8, and so on.
Subshells are sets of states within a shell, defined by the azimuthal quantum number ℓ. ℓ = 0, 1, 2, 3 correspond to orbitals s, p, d, f.
| Atomic Mass | Atomic Number |
|---|---|
| Total number of protons and neutrons in the nucleus | Number of protons in the nucleus (atomic number calculator) |
| Represents average mass of an atom | Determines position in periodic table |
| Denoted by A | Denoted by Z |
| Measured in atomic mass units (AMU) | Whole number used to identify elements |
Electron configuration follows the Pauli Exclusion Principle, Aufbau Principle, and Hund's Rule. Use these rules to determine how electrons occupy orbitals.
K, L, M, N are electron energy levels: K = 1st shell, L = 2nd shell, M = 3rd shell, N = 4th shell. This notation shows how many electrons occupy each shell.
Electrons have negligible mass (~9.11 × 10⁻²⁸ g), so they do not significantly affect atomic mass.
Mnemonic: "Happy Henry Lives Beside Boron Cottage"
The noble gas electron configuration calculator provides a simple way to find condensed electron configurations, atomic number, and atomic mass. The free orbital diagram calculator quickly shows how many orbitals an atom has and how electrons are distributed.
Related
Links
Home Conversion Calculator About Calculator Online Blog Hire Us Knowledge Base Sitemap Sitemap TwoEmail us at
Contact Us© Copyrights 2026 by Calculator-Online.net