Calculator Online
Pages
Category
Follow Us On:
Enter the number of valence electrons, number of lone and bonded pairs. The calculator will readily determine the formal charge.
Related
An online formal charge calculator is designed to calculate the formal charge of an atom. Understanding formal charge is important before using this free tool to ensure accurate results.
“The individual charge of each atom present in a molecule.”
Formal charge indicates the reactivity of a molecule and how it will behave when forming bonds with other atoms.
The Lewis structure shows the bonding electron pairs of atoms and the lone pairs in a molecule.
Carbon dioxide (CO2):
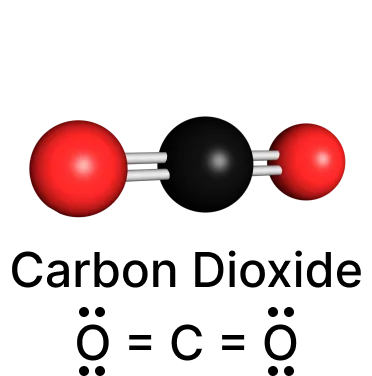
Sulfur dichloride (SCl2):
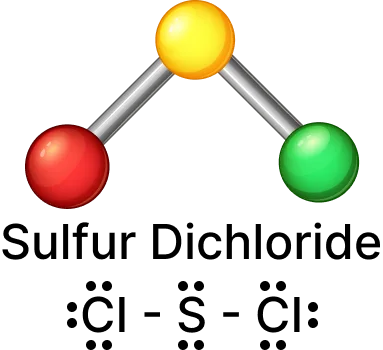
“A pair of electrons that does not participate in bonding is called a lone pair.”
Example – Nitrogen Trichloride (NCl3): Bond pairs shown as -, lone pairs as :
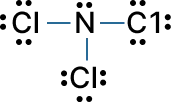
To calculate formal charge:
FC = V - (LP + 0.5 × BE)
Lewis structure of SO2:

Formal charge:
FC = 6 - (2 + 0.5 × 8) = 6 - 6 = 0
Lewis dot structure of F:
![]()
FC = 7 - (6 + 0.5 × 2) = 7 - 7 = 0
Lewis structure of CH3O-:
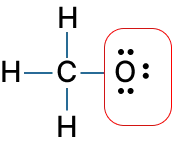
FC = 6 - (6 + 0.5 × 2) = 6 - 7 = -1
Input:
Output:
Electrons shared among atoms must be balanced to determine accurate Lewis structures.
A negative formal charge occurs when an atom gains extra electrons to complete its octet.
No, they are theoretical values used to track electron distribution.
A formal charge of 0 is ideal, indicating fully filled electron shells during bonding.
No, formal charge does not directly determine molecular polarity.
Yes, formal charges indicate regions of electron density, which influence the dipole moment.
Formal charge is crucial for predicting reaction mechanisms and tracking electron sharing among atoms. An online formal charge calculator ensures precise results quickly, minimizing errors in chemical analysis.
Related
Links
Home Conversion Calculator About Calculator Online Blog Hire Us Knowledge Base Sitemap Sitemap TwoEmail us at
Contact Us© Copyrights 2026 by Calculator-Online.net