Calculator Online
Pages
Category
Follow Us On:
Enter the chemical equation and the calculator will immediately let you know which element is oxidised and which is reduced in it.
Related
The redox reaction calculator identifies both oxidation and reduction simultaneously, making it easy to determine oxidizing and reducing agents. Some ions are naturally oxidizing, while others act as reducing agents.
A redox reaction is a chemical reaction in which oxidation and reduction occur at the same time. It can be challenging to identify where each process occurs, but a redox reaction balancer automatically shows the changes and identifies the reducing and oxidizing agents.
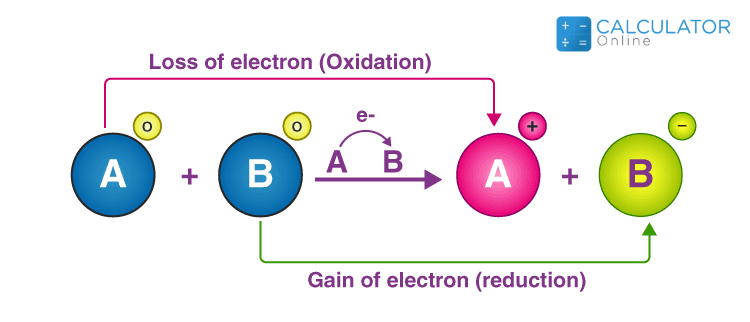
Reduction involves specific changes in atoms or ions:
Reaction: 2FeCl3 (aq) + H2 (g) → 2FeCl2 (aq) + 2HCl (aq)
FeCl3 is reduced as it gains electrons; Hydrogen is oxidized as it loses electrons.
Oxidation is the reverse of reduction:
Reaction: Mg (s) + O2 (g) → 2MgO
Magnesium is oxidized as it loses electrons; Oxygen is reduced as it gains electrons.
Reaction: H2 + F2 → 2HF
Reaction: Zn (s) + CuSO4 (aq) → ZnSO4 (aq) + Cu (s)
Input: Enter the chemical equation in the calculator.
Output:
What are oxidizing agents?
Oxidizing agents accept electrons and are reduced in the reaction. Examples: O2, O3, H2O2, F2, HNO3.
What are reducing agents?
Reducing agents donate electrons and are oxidized. Examples: Mg, Li, Br, Fe.
Are metals reducing agents?
Yes, metals tend to lose electrons. Examples: Fe, Mg, Na.
Are nonmetals oxidizing agents?
Yes, nonmetals tend to gain electrons (except hydrogen). Examples: O2, F2.
Redox reactions involve simultaneous oxidation and reduction. A redox reaction calculator simplifies understanding by clearly identifying electron transfer and balancing equations.
Related
Links
Home Conversion Calculator About Calculator Online Blog Hire Us Knowledge Base Sitemap Sitemap TwoEmail us at
Contact Us© Copyrights 2026 by Calculator-Online.net