Calculator Online
Pages
Category
Follow Us On:
Enter the values to calculate the rate of change and the rate of reaction in dynamic processes.
Related
Use the rate constant calculator to determine the rate constant and reaction rate for a given substance over a specified period. It simplifies the calculation of reaction kinetics.
The rate constant indicates how fast or slow a reaction occurs. It depends on temperature and activation energy. Higher temperature increases k, while higher activation energy decreases it.
Reaction rate is influenced by reactant concentration, temperature, catalysts, and surface area.
kt = [R₀] – [R]
k = ([R₀] – [R]) / t
Initial concentration of A ([A₀]) = 0.2 M, after 30 minutes [A] = 0.1 M. Find k for a first-order reaction.
For a first-order reaction:
[A] = [A₀] · e^(-kt)
Step 1: Substitute values: 0.1 = 0.2 · e^(-k·30)
Step 2: Divide both sides by 0.2: e^(-k·30) = 0.5
Step 3: Take natural logarithm: -k·30 = ln(0.5)
Step 4: Solve for k: k = -ln(0.5)/30
This gives the first-order rate constant.
Reaction rate can follow Zero, First, or Second-order kinetics.
Rate is independent of reactant concentration. Half-life: t₁/₂ = [A₀] / (2 × k)
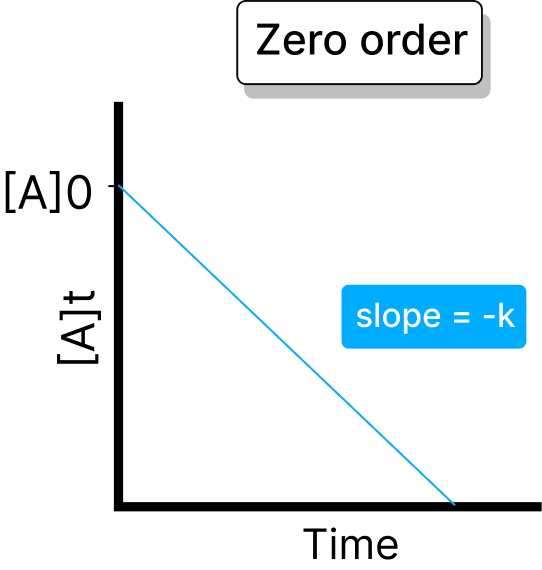
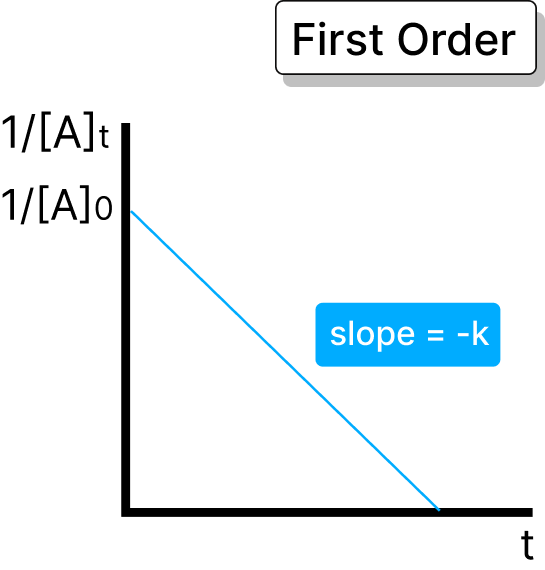
Rate is directly proportional to reactant concentration. Half-life: t₁/₂ = 0.693 / k
Rate = k × [A]
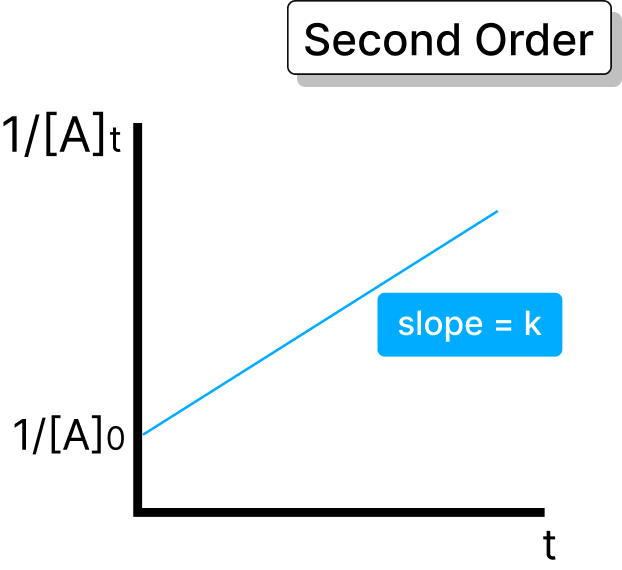
Rate ∝ square of one reactant or product of two reactants. Half-life: t₁/₂ = 1 / (k × [A])
Half-life is the time required for a substance to decrease by half. In radioactive decay, it's the time for half of a substance to decay. For drugs, it may take ~5 half-lives to be cleared.
Depends on reaction order. Example: L·mol⁻¹·s⁻¹ for second-order reactions.
Higher temperature generally increases the rate constant, while lower temperature decreases it.
No, k is always positive as it represents the speed of concentration change over time.
Related
Links
Home Conversion Calculator About Calculator Online Blog Hire Us Knowledge Base Sitemap Sitemap TwoEmail us at
Contact Us© Copyrights 2026 by Calculator-Online.net